What is a Rechargeable Lithium Battery and How Does it Work?
Rechargeable Lithium Batteries are a cornerstone of modern energy storage. These batteries have revolutionized technology, powering everything from smartphones to electric vehicles. According to the International Energy Agency, the global demand for lithium-ion batteries is projected to reach 11 terawatt-hours by 2030. This growth reflects an increasing reliance on renewable energy sources and efficient storage solutions.
Their unique chemistry allows them to charge and discharge efficiently. This efficiency shapes the renewable energy landscape, enabling longer-lasting devices and reduced carbon footprints. However, challenges persist in battery recycling and sustainability. Current recycling rates for lithium batteries remain low, highlighting a need for improvement.
As the market expands, so do concerns over resource extraction and environmental impact. Striking a balance between innovation and responsible sourcing is crucial. Rechargeable Lithium Batteries symbolize advancements, yet they also prompt reflection on their lifecycle and ecological footprint. Addressing these complexities will define the future of energy storage.

What is a Rechargeable Lithium Battery?
A rechargeable lithium battery is a type of battery that can be used multiple times. These batteries are widely known for their effectiveness in powering devices like smartphones and laptops. They can be charged using electricity, which makes them convenient for daily use. When you plug in your device, the battery stores energy for later use.
The structure of a lithium battery plays a crucial role in its functionality. Typically, it consists of an anode, cathode, and electrolyte. The anode often contains graphite, while the cathode uses lithium compounds. When the battery discharges, lithium ions move from the anode to the cathode. Conversely, during charging, the process reverses.
While lithium batteries are praised for their long life, they are not perfect. They can degrade over time, reducing their efficiency. Improper charging might lead to safety issues. Additionally, their production has environmental consequences. However, they remain a popular choice due to their lightweight nature and high energy density. This adds to the challenge of balancing performance and sustainability.
What is a Rechargeable Lithium Battery and How Does it Work?
| Feature | Description |
|---|---|
| Chemistry | Lithium-ion and Lithium-polymer |
| Cycle Life | 300 to 500 charge cycles |
| Energy Density | 150-200 Wh/kg |
| Charging Time | 1 to 4 hours depending on capacity |
| Voltage | Typically 3.6 - 3.7 volts per cell |
| Self-discharge Rate | About 5% per month |
| Common Applications | Smartphones, laptops, electric vehicles, and power tools |
| Safety Features | Built-in thermal protection, overcharge protection, and short-circuit prevention |
Components of a Lithium-Ion Battery Explained
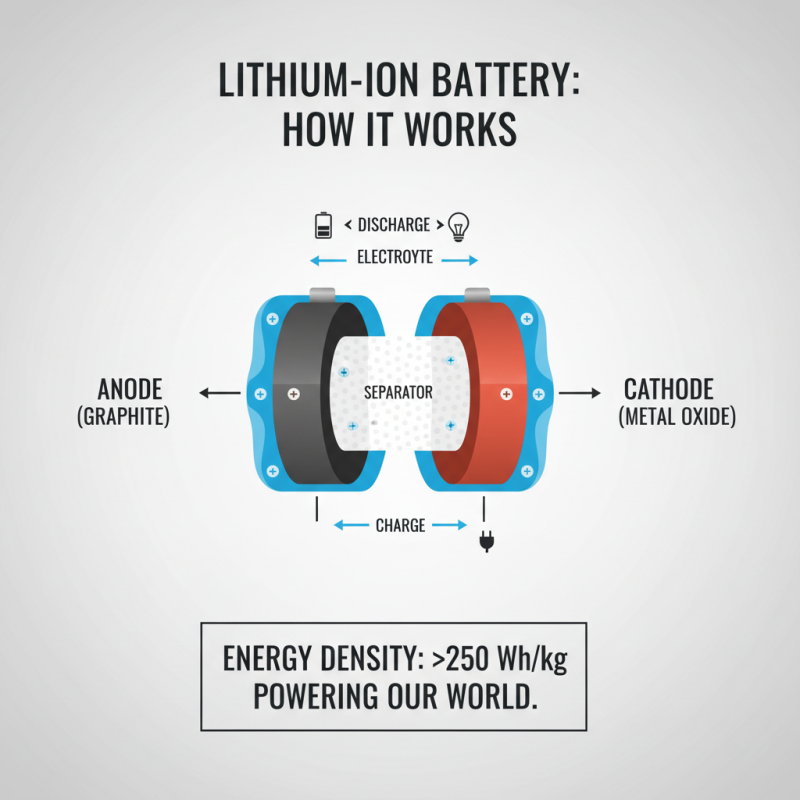
Lithium-ion batteries are widely used today. Their construction involves several key components that work together for effective energy storage. The core components include the anode, cathode, electrolyte, and separator. The anode typically consists of graphite, while the cathode can be made of lithium metal oxides. This combination allows for significant energy density, with some estimates suggesting that lithium-ion batteries can offer over 250 Wh/kg of energy.
The electrolyte is crucial for ion transport. It conducts lithium ions between the anode and cathode during charging and discharging. However, the choice of electrolyte can affect battery safety and performance. A liquid electrolyte is common, but solid-state options are emerging. Interestingly, researchers are exploring alternatives to traditional materials. For example, silicon-based anodes offer a boost in capacity, potentially doubling charge storage compared to graphite.
Despite advancements, lithium-ion technology has limitations. For instance, battery life can diminish over time. A study indicated that energy capacity degrades by about 20% after 500 cycles. Additionally, environmental concerns regarding lithium extraction and recycling remain pressing. The industry must address these issues to improve sustainability. The evolution of battery technology will depend on innovation and a commitment to responsible practices.
How Charging and Discharging Works in Lithium Batteries
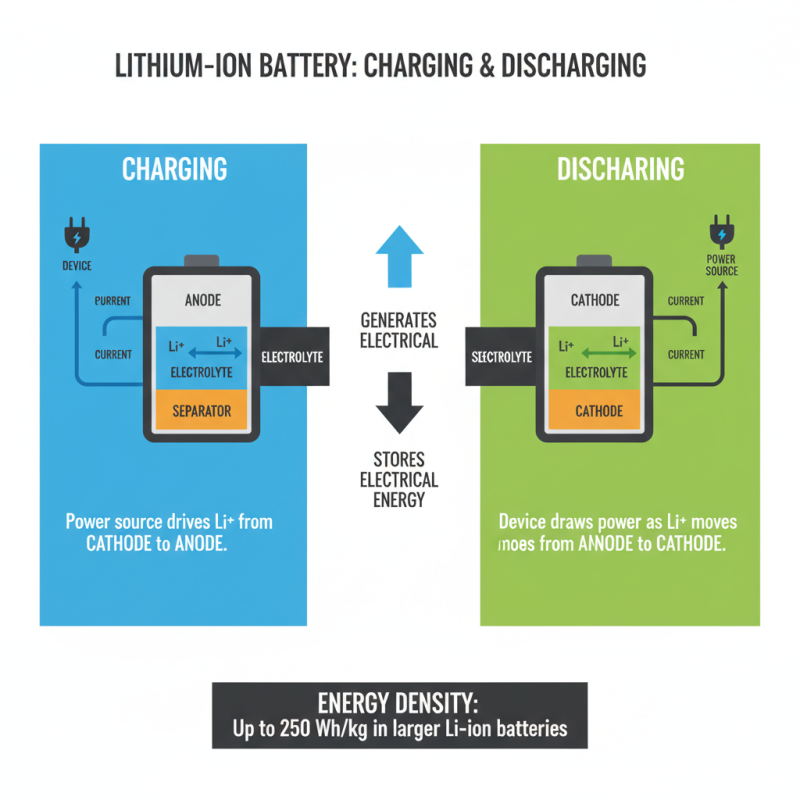
Charging and discharging in lithium batteries is a fascinating process. Lithium ions move between the anode and cathode during these cycles. This movement generates electrical energy. When you plug in a device, the charging process begins. Current drives lithium ions from the cathode to the anode. This cessation creates a balance. Larger lithium-ion batteries can store up to 250 Wh/kg, according to recent reports.
Discharging is the opposite. When a device is in use, lithium ions flow back to the cathode. This process releases energy, powering our electronics. Reports indicate that about 80% of the energy transferred can be used effectively. However, some energy is always lost as heat during this transition. This inefficiency can be problematic for high-drain devices. In some cases, battery life may decrease by about 20% after numerous charge cycles.
Many factors influence this system. Temperature, charge rates, and battery age play crucial roles. For instance, charging at high speeds can degrade battery life quickly. This complexity leads to ongoing research in battery technology. Improved longevity and efficiency remain key goals. The quest for better solutions continues.
Advantages of Using Rechargeable Lithium Batteries
Rechargeable lithium batteries have become a common choice for many devices. Their advantages are compelling. These batteries are often lighter than other types. This makes devices more portable. Moreover, they can hold a significant amount of energy. This aspect is particularly beneficial for smartphones and laptops.
One notable advantage is the long lifespan. A good lithium battery can last for years if maintained properly. It's crucial to avoid complete discharges frequently. This can lead to reduced capacity over time. Another key point is efficiency. These batteries charge quickly, making them convenient. For example, a short charging session can provide hours of use.
However, there are considerations to keep in mind. They can be sensitive to heat, which can cause issues. Storing them in a hot environment isn't wise. Additionally, improper disposal can harm the environment. Awareness of recycling options is essential. Balancing longevity and eco-friendliness is a challenge for users.
Applications of Rechargeable Lithium Batteries in Modern Technology
Rechargeable lithium batteries are changing how we use technology today. These batteries are commonly found in smartphones, laptops, and electric vehicles. Their lightweight design and high energy density make them ideal for portable devices. Users enjoy longer usage times without the hassle of frequent recharging. However, the production and disposal still raise environmental concerns.
In addition to consumer electronics, lithium batteries also play a significant role in renewable energy. They store energy generated from solar panels and wind turbines. This stored energy ensures a steady power supply, even when the sun isn’t shining or the wind isn’t blowing. Many households are adopting these systems, helping reduce reliance on fossil fuels. Yet, the cost and complexity of installation can be barriers for some.
Despite their advantages, rechargeable lithium batteries have limitations. Over time, their performance can degrade, leading to shorter usage periods. Further, the mining of lithium poses ethical challenges related to labor practices. Innovations in recycling methods are necessary to mitigate these issues and improve sustainability. Balancing the benefits and drawbacks is vital for the future of battery technology.
Related Posts
-

Why Are Lithium Batteries Essential for Modern Technology and Sustainability
-

Why Are Lithium Batteries So Important for Our Future Energy Needs
-
2025 Top Lithium Car Battery Innovations for Electric Vehicle Efficiency
-

Top Reasons to Choose Lithium RV Battery for Your Next Adventure?
-

Ultimate Guide to Choosing the Best Rechargeable Lithium Batteries for Your Devices: Expert Insights & Data
-

Exploring Alternative Energy Solutions with Lithium Metal Batteries
